Creating Chemosensing Material from a Discrete Macrocyclic Complex
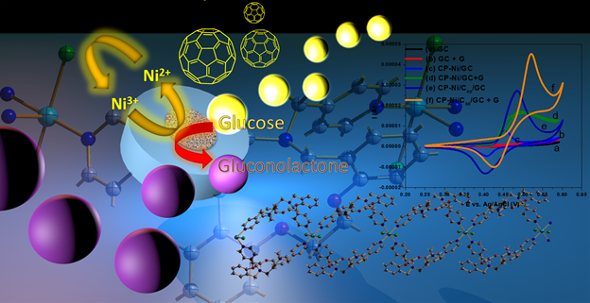
Nickel(II) coordination polymer Ni(II)-CP was synthesized within a series of 1D CPs by using diaza-macrocycles, bearing pyridine side arms via an incredible easy method at room temperature without using of solvothermal, ultrasonic and mechano-chemical methods. Further observations revealed that Ni(II)-CP enjoyed from excellent electrochemical properties in terms of glucose sensing, capable to act as a non-enzymatic glucose sensor for its crystalline layered structure. These observations resulted in a novel modified glassy carbon electrode employing Ni(II)-CP/C60 composite for non-enzymatic detection of glucose, wherein Ni (II)-CP/C60/GCE exhibited remarkable sensing properties as high sensitivity, low detection limit, wide linear range as well as excellent selectivity.
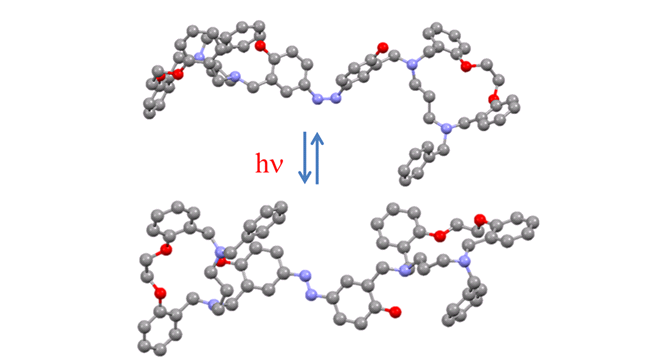
Molecular Photo-Switch
Among the photoactive functional groups, azobenzenes have been explored for decades as influential molecular switches and have been employed in numerous different areas stretching from liquid crystals to biomolecules and switchable catalysts. Fundamentally, the azobenzene moieties can undertake reversible photo-isomerization between the over-extended trans (E-isomer) and the twisted cis (Z-isomer) isomers upon exposure to light at a certain absorbed wavelength or heating, accompanied with substantial alterations in the shape of the molecule.
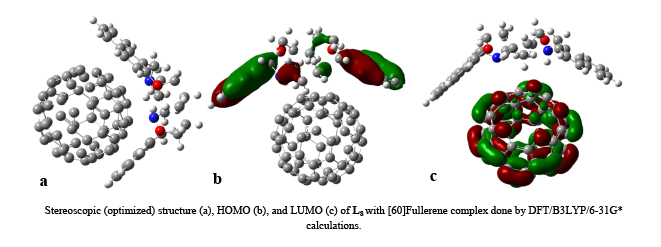
Complexation of Naphthalene Azacrown Macrocyclic Ligands with [60]Fullerene and Metal Cations
A series of 15- to 19-membered N2O2-donor naphthodiaza-crown macrocyclic ligands with intrinsic emission properties synthesized.After synthesis and charaterization of mentioned ligands, their complexation with [60]Fullerenes and metal cations investigated. The fluorescence behaviors of these ligands in the presence or absence of [60]Fullerenes and metal cations studied.
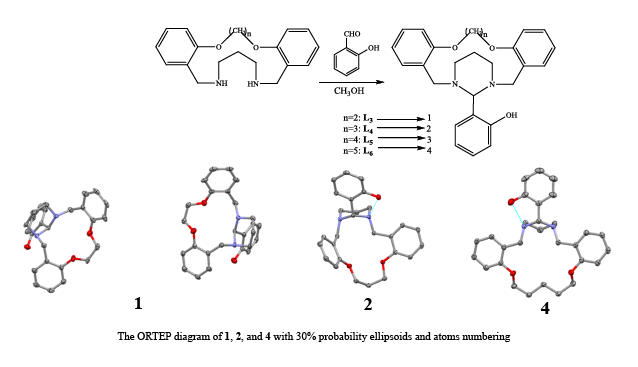
Synthesis and Characterization of a New Group of Exo-Coordinating O2N2-Donor Macrocycles
The reaction of the 15–18 membered benzo-diaza-crown ethers with salicylaldehyde afforded an n-membered O2N2-donor macrocyclic ligands mounted with 1,3 diazacyclohexane sub-rings, 1-4, in high yield.
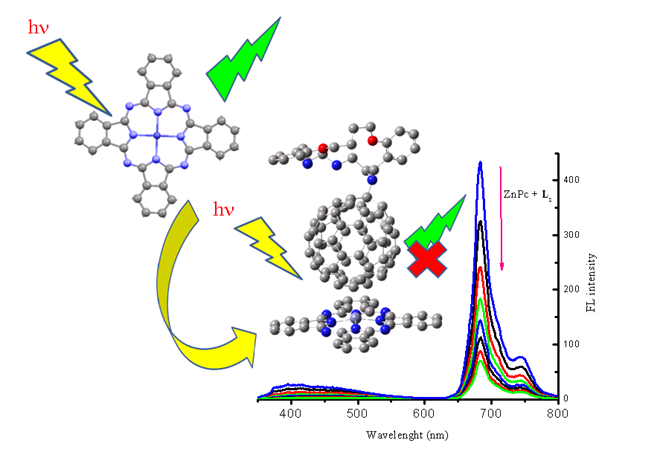
Through-Space Electronic Communication of Zinc Phthalocyanine with Substituted [60]Fullerene
The ground state non-covalent interaction of [60]Fullerene derivatives of O2Nx (x = 2, and 3) aza-crown macrocyclic ligands with zinc phthalocyanine (ZnPc) was also investigated by UV–vis absorption, steady state and time resolved fluorescence spectrophotometry.
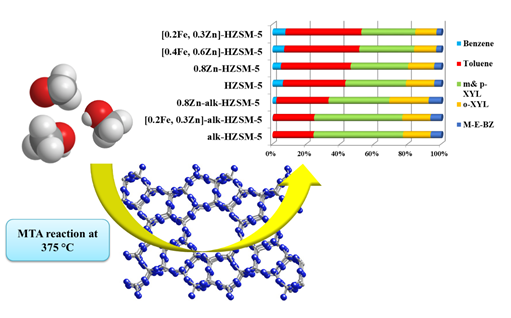
Conversion of methanol to hydrocarbons (MTH)
The methanol is supplied from different resources as syngas, natural gas, coal, biomass and it can be converted into hydrocarbons, such as ethene and propene olefins (MTO and MTP), high octane gasoline (MTG) and aromatics (MTA) over acidic zeolite or zeolite-type catalysts by the methanol-to-hydrocarbon (MTH) process.
The conversion of methanol to aromatic hydrocarbons (MTA) was studied over a new series of mesoporous low-silica HZSM-5 catalysts. The catalysts were prepared in the absence and presence of the promotional effect such as Zn and Fe in both alkaline and neural aqueous solutions.